In recent years, traditional Chinese medicine has become an increasingly important aspect of healthcare in Richmond. This form of medicine, which has been used for thousands of years in China, has been gaining more and more popularity among Westerners. While many people remain skeptical about its effectiveness, Chinese Medicine Richmond has been proven to be a viable alternative to Western medicine.
One of the core beliefs of Chinese medicine is that the human body is a self-healing organism. Thus, the goal of traditional Chinese medicine is to promote and facilitate the body’s natural healing capabilities. Unlike Western medicine, which primarily focuses on curing the symptoms of an illness, Chinese medicine aims to treat the root cause of the problem.
There are several different modalities that fall under the umbrella of traditional Chinese medicine. Some of the most popular include acupuncture, herbal medicine, and cupping therapy. Each of these modalities relies on different techniques to help the body heal itself.
Acupuncture is perhaps the most well-known aspect of traditional Chinese medicine. This modality involves the insertion of small needles into specific points on the body. These needles stimulate the flow of energy, or qi, throughout the body. According to Chinese medicine, when the qi is moving smoothly, the body is healthy. However, when the qi becomes blocked or stagnant, illness and disease can occur.
Herbal medicine is another common aspect of Chinese medicine. Practitioners of traditional Chinese medicine use a variety of herbs to treat different ailments. Each herb has its own unique properties, and practitioners often use several different herbs in combination to create a formula tailored to each patient’s specific needs.
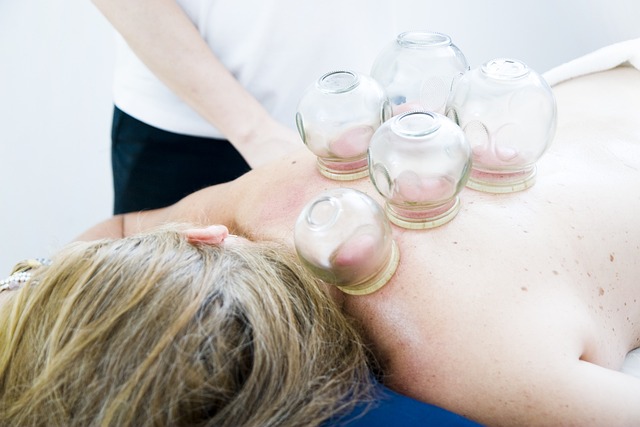
Cupping therapy is a third modality that falls under the umbrella of Chinese medicine. This technique involves the application of cups to the skin, which creates a vacuum. This suction increases blood flow to the area, which can help to relieve pain and promote healing.
While traditional Chinese medicine may seem foreign to many Westerners, it has been proven effective in treating a variety of conditions. Studies have shown that acupuncture can help with conditions such as chronic pain, headaches, and even depression. Herbal medicine has been used to treat everything from the common cold to cancer. And cupping therapy has been shown to reduce pain and inflammation.
One of the benefits of traditional Chinese medicine is that it can be used in conjunction with Western medicine. While Western medicine can be effective in treating acute conditions, it often falls short when it comes to chronic conditions. Traditional Chinese medicine can provide a complementary approach to treating chronic conditions, such as arthritis or fibromyalgia. By working together, Western medicine and Chinese medicine can provide a more holistic approach to healthcare.
In addition to its effectiveness, traditional Chinese medicine is also generally considered safe. While there may be some minor side effects associated with different modalities, such as bruising from acupuncture or upset stomach from herbal medicine, these side effects are generally mild and short-lived.
Chinese Medicine Richmond offers a holistic, natural approach to healthcare that has been used successfully for thousands of years. While Western medicine has its place, traditional Chinese medicine can provide a complementary approach that addresses the underlying causes of illness and promotes the body’s natural healing capabilities.







